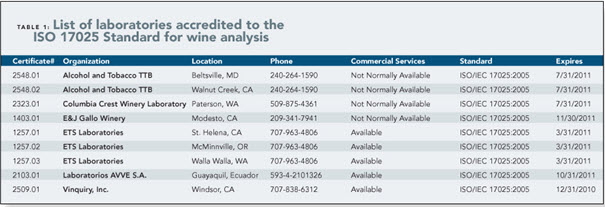
Testing facilities and laboratories seeking accreditation to meet a specific customer’s needs, to enhance marketability, or satisfy the requirements of a regulatory agency would be well-advised to become accredited by ISO 17025 standards. ISO17025 is not the only method of accreditation, but it does provide a level of visibility that few other programs offer because it is an internationally-recognized standard. Companies who are looking to contract out testing services very often look for ISO17025 accreditation for this very reason.
ISO 17025 provides a roadmap of the general requirements needed by testing and calibration laboratories to prove competency. The standard applies to any lab which does sampling, testing and calibration regardless of the number of employees at the facility or the type of work that will be undertaken. ISO 17025 accreditation is possible whether the lab does testing by standard procedures, non-standard procedures, or its own lab-generated procedures. The standard does not verify the validity of a test, it merely provides the structure that a facility can use to develop, document, and maintain managerial and technical quality programs.
Testing and calibration processes do not have to occur at any specific point in order for a laboratory to become accredited. Internal, external, or third-party labs can be accredited if they use these procedures as part of a product certification, or if their output is solely a test or calibration result. Accreditation can also be granted to any lab that performs sampling as its primary role.
ISO 17025 is broken down into five sections:
- The Scope of the Standard,
- Normative References,
- Terms and Definitions,
- Management Requirements, and
- Technical Requirements.
The substance of the standard really lies within the last two sections, which define how the lab management develops, controls, and maintains a quality management system, and how reliable, repeatable, and correct the tests performed by the lab actually are. The last revision of the standard was issued in 2005, and the main changes between it and its predecessor dealt primarily with the increased involvement and continual improvement efforts of the senior management team.
In order for testing and calibration laboratories to become accredited, they must have in place a quality management system that is defined, documented, and followed. This is the purpose of ISO 17025: To provide the outline for a facility to develop and implement that management system. A quality control manual, documented procedures and controls, management oversight and employee involvement are all present in this system as they are every other ISO-driven quality management system. Once the process and procedures are in place, the lab must implement the quality management plan and prove compliance with the system for many months before the process of accreditation can begin.
from WineBusiness.com
Figure: Becoming Accredited Can Provide Great Benefits to a Lab Seeking to Expand its Business Reach
When a lab has documented long-term compliance with their self-generated quality control system, they can request accreditation from a certified third-party accreditation agency which has jurisdiction over that part of the world. The accreditation body will review the lab’s quality management program, including compliance, records and records maintenance, and the consistency and accuracy of the test results being output. If the assessment is successful, the lab will be granted accreditation under ISO 17025. When a lab becomes accredited, it means it has proven competence in the realm of testing and calibration, has demonstrated consistent results and the reporting of those results, and can back that up with an active system of continuous improvement for the management system, employee involvement, and compliance to its own quality control program.
It is important to point out here that accreditation is not the same thing as certification. Although ISO 17025 and ISO 9001 are similar, they are not identical, nor are they meant to be. ISO 17025 provides a roadmap for labs to build and implement quality management programs, which is only one of many requirements for ISO 9001 certification. In most instances, being accredited under ISO 17025 provides enough credibility on its own to help a lab expand its reach into the business community, but it merely denotes competency, not accuracy.
back to ISO Standards